Molecular point-of-care Testing (POCT)
A point-of-care test (POCT) is a laboratory test performed
near the patient's point of care. POCT provides rapid test results and can
improve patient care. However, if performed incorrectly or used incorrectly,
POCT can produce misleading results and require additional monitoring,
increasing costs and risk to patients. Although most POCTs were eliminated in
the Federal Clinical Laboratory Improvement Amendments of 1988, several
considerations challenge their implementation. This chapter will focus on the advantages
and limitations of POCT and describe how to manage the quality of POCT to
achieve optimal patient outcomes.
What are the point-of-care devices for under-detection of drug abuse?
Point-of-care testing (POCT) devices are available at the bedside or in the doctor's office to test for drugs using a urine sample or oral fluid.
The advantage of this test is the quick availability of results. The POCT unit
uses immunoassay and lateral flow chromatography principles.
Urine drug screening generally uses cutoff concentrations for various drugs prescribed by SAMHSA (Substance Abuse and Mental Health Services Administration), but there are exceptions.
SAMHSA's mandatory guidelines can also be used to test oral fluids. However, POCT devices may not be as reliable as laboratory drug tests due to a higher rate of false positives and some false negatives.
However, POCT devices have gained recognition for drug testing in
urine and oral fluids due to their simple operation and rapid availability of
results.
How to monitor oxygen status?
Although part of a common
"blood gas" test panel with pH and pCO2, pO2, %O2Hb and related
parameters can be used independently to detect and monitor oxygen loss from a
variety of causes.
Blood gas and oxygen saturation measurements can be performed using laboratory analyzers, test points, noninvasive pulse oximetry, and transcutaneous blood gases.
The type and condition of specimen selected for oxygen monitoring may be based on a combination of urgency, practicality, clinical need, and treatment goals.
Because blood oxygen concentrations are extremely unstable, some very important pre-analysis steps must be taken to prevent errors in oxygen and oxygen saturation results. Effective use of oxygen requires binding to hemoglobin in the lungs, transport in the blood, and release into the tissues where cellular respiration takes place.
Hydrogen ions (pH), CO2, temperature and 2,3-DPG play important roles in these processes.
Additional measurements and calculations are often used to explain and determine the cause and source of hypoxia. These include Hb concentration, alveolar-arterial pO2 gradient, pO2:FIO2 ratio, oxygenation index, O2 content and O2 delivery, as well as pulmonary dead space and intrapulmonary shunting.
The causes of hypoxemia will be discussed and two clinical cases will be
presented and described to illustrate how oxygen parameters are used clinically
in the diagnosis and management of patients with abnormal oxygenation.
What is the point-of-care model for critical care analytics?
Point-of-care testing (POCT), specifically blood gas and electrolyte testing,
is now ubiquitous in many patient and critical care settings.
To provide a practical example of the challenges of providing POCT, a case example of implementing POCT in an ICU setting is presented.
Point-of-care testing (POCT) is now common in many close patient and critical care settings.
For blood gas and electrolyte testing, this includes the operating room (OR), intensive care unit (ICU), cardiac catheterization laboratory (CCL), emergency department (ED), and many primary care clinics.
POCT has several distinct advantages, such as the reduction or elimination of
sample transport and handling, with the added benefit of reduced pre-analytical
impact and faster turnaround time (TAT).
Some systems offer a series of test boxes that provide a
flexible test menu. Faster test results can allow faster medical decisions,
leading to improved patient outcomes, operational efficiency and patient
satisfaction.
For a variety of reasons, POCT devices typically require less blood, especially compared to central laboratory requirements, and are particularly attractive in pediatrics.
Because POCT is often not performed by trained laboratory personnel, maintaining regulatory compliance and quality assurance using POCT is a challenge that requires continuous monitoring.
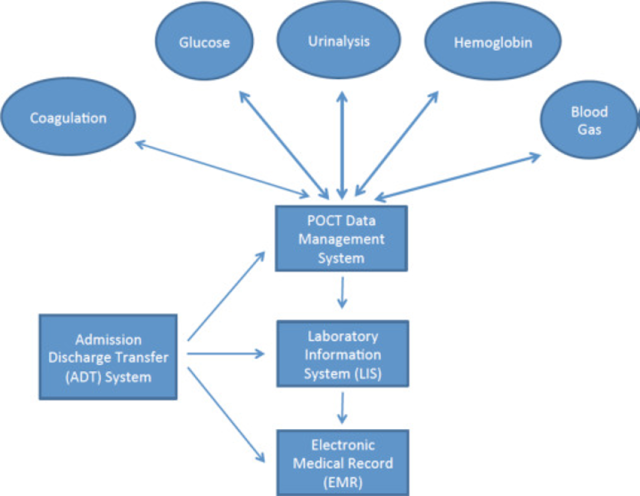
Most current POCT devices can be connected to laboratory information systems (LIS) and electronic medical record (EMR) systems.
Given today's high volume of
testing and the need to minimize transcription errors, connectivity to
information systems is essential.
A well-functioning information system can automatically transfer (download) the results from the analyzer and display them to the doctor. It ensures accurate and fast delivery of results to multiple relatives. An issue that sometimes arises is the potential for POCT to replace central laboratory (CL) testing.
Can POCT replace CL blood tests?
POCT has and will replace some CL tests, and POCT accounts for 10-20% of clinical laboratory tests, depending on location. This applies to situations where quick results are essential for emergency decision-making, and is useful in non-emergency situations such as clinics, which can allow doctors to assess results and discuss them with patients while they are still there.
In addition, POCT can replace extremely inefficient near-patient laboratories. However, the high volume of tests performed by most central laboratories cannot be replaced by POCT, at least with current technology.
Most, if not all, POCT units are not equipped to perform a large
number of tests and are often much more expensive to operate.
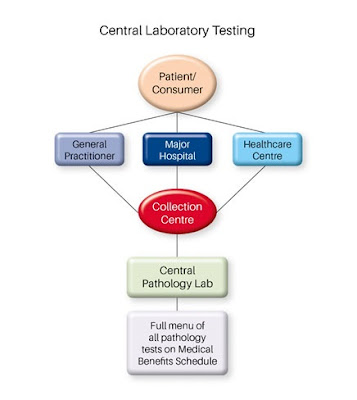
Laboratory Organization, Management and Security
Point-of-care testing (POCT), also known as next-to-patient testing, refers to any analytical test that is performed outside of a laboratory, possibly in a hospital as an adjunct to a main laboratory or for primary care outside of a hospital setting.
The users of this service are usually not laboratory staff. Public awareness of the availability of POCT has increased dramatically, and access to the technology has never been greater.
If results need to be used to make rapid clinical decisions, laboratory testing in the clinical setting may be the best option for patients.
Diagnostic laboratories are often located in areas of the hospital away from intensive care and outpatient areas.
Rapid transport systems, including pneumatic tubes
(see above), may be a desirable alternative to multi-satellite test areas,
especially if the main laboratory is already providing rapid results.
Laboratory test turnaround times (see above) must be known to make an informed decision as to whether close monitoring of the patient in the satellite area is warranted.
If POCT equipment is preferred, the operation of
the satellite laboratories and the maintenance of the equipment should be the
responsibility of the relevant pathology discipline.
The hematology laboratory manager or specialist POCT
coordinator should have overall responsibility for this service. This is
essential for quality control, safety and certification, whether the satellites
are operated by laboratory staff, for example in busy areas, or used by medical
staff or nurses as ancillary activities.
Designated laboratory personnel must monitor this service, visit each test site daily, and ensure that all results and quality control data are integrated into the laboratory's main computer system.
Guidelines for the
organization of POCT services have been published by the British Hematology
Standards Board 33 and the UK Medical Devices Agency 34 , the US Society for
Clinical and Laboratory Standards 35 and the International Committee for
Standardization in Hematology 36 and internationally. International
Organization for Standardization.
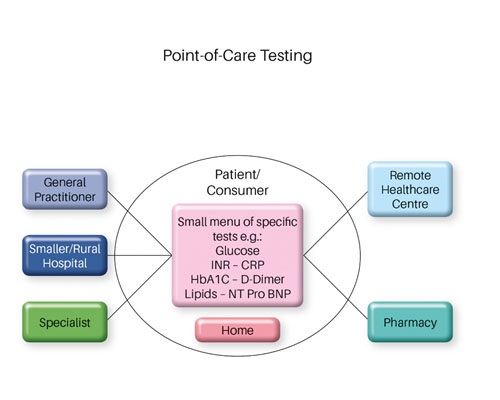
What is Point-of-care Testing outside the Laboratory?
Out-of-laboratory POCT is becoming increasingly popular in
some countries and is particularly useful when patients are located far from
hospital laboratories. Instrument manufacturers now produce tabletop or
handheld devices that are easy to use, self-calibrate, and require minimal
maintenance.
Common hematologic tests include hemoglobin concentration, blood count using a simple analyzer, erythrocyte sedimentation rate, and prothrombin time to monitor oral anticoagulation.
Although this use of POCT is
autonomous, local hospital laboratories should encourage physicians and clinics
to seek advice and assistance in selecting appropriate instruments and in their
standardization/calibration and quality control, including links to external
quality assessment programs in which the laboratories participate.
Reconciliation of reports with laboratory records is useful
when patients are referred to the hospital. Research on anticoagulation control
management suggests that with appropriate training and laboratory
collaboration, pharmacists can provide services as reliable as hospital
services and generally more convenient for patients. 38
A major source of error in out-of-hospital POCT is sample
collection errors, whether it is a vein sample or a finger sample. Supervised
training should be given to the clinic staff engaged in this specialization.
What are some Developments in point-of-care cardiovascular disease screening?
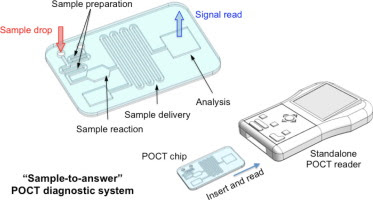
Point-of-care testing (POCT), commonly referred to as near-patient testing or point-of-care testing, is a diagnostic alternative to laboratory analysis.
Typically, POCT is performed close to the patient and
results are obtained in a short period of time (ideally < 20 minutes).
This rapid format is less resource-intensive than laboratory testing and
focuses on the availability of highly advanced point-of-care (POC) devices to
allow detection of specific biomarkers in a robust and simplified manner.
These devices may take the form of a single disposable element or may contain an entire platform into which the disposable insert can be placed. These platforms usually consist of dedicated control and readout systems and include disposable cartridges for test loading.
In addition, custom laboratory analysis elements and custom structures are often packaged into these kits in compact and highly coherent configurations to facilitate automated sample analysis.
This automation is essential to the versatility of
POC devices and their ability to support simplified biomarker measurements in
complex samples.
To some extent, these platforms also incorporate error
handling in their design to minimize error handling. This aspect is influential
in reducing the need for highly specialized training in the use of these
devices compared to the operation of complex laboratory instruments.
In addition, these devices can be used in a variety of
settings, further improving the identification and ongoing management of CVD.
Author's Bio
Education: MBBS, MD
Occupation: Medical Doctor
Specialization: Community Medicine, General Surgery, Natural Treatment
Experience: 18 Years as a Medical Practitioner
Share this in Public Information Interest>>



Comments
Post a Comment