Types and Treatment of Hypoxia
Hypoxia refers to a pathological process in which the
metabolism, function and morphological structure of tissues are abnormally
changed due to insufficient oxygen supply or oxygen use disorders in tissues.
Hypoxia is a very common pathological process in various clinical diseases.
Hypoxia in vital organs such as brain and heart is also an important cause of
death. In addition, due to the significant decrease in arterial blood oxygen
content, the tissue oxygen supply is insufficient, also known as hypoxemia.
Hypoxia refers to the lack of endogenous oxygen, which is
oxygen bound to cells.
Hypoxia refers to a type of pathological process that is very common in hypoxic diseases due to insufficient oxygen supply or oxygen use disorder to tissues.
The significant
reduction of arterial blood oxygen content has a significant biological
process.
|
Table of Contents |
|
1. Introduction to Indicators 2. Type Features 3. Functional Changes 4. Metabolic Changes 5. Treatments
|
Indicator Introduction
The uptake and utilization of oxygen by the body is a complex biological process. Generally speaking, to judge the state of oxygen acquisition and utilization by tissue, two factors should be detected: the oxygen supply to the tissue and the oxygen consumption of the tissue.
Determination of blood oxygen parameters is necessary to understand the
acquisition and consumption of oxygen in the body:
1.1 Partial pressure of oxygen (P O2 )
The tension created by the oxygen physically dissolved in the blood. The partial pressure of oxygen in arterial blood (Pa O2) is about 13.3kPa (100mmHg), and the partial pressure of oxygen in venous blood (Pv O2) is about 5.32kPa (40mmHg).
At the same time, it is also the dynamic factor of oxygen
diffusion to tissues; while PvO2 reflects the state of internal respiratory
function.
1.2 Oxygen binding capacity (CO2max)
CO2max refers to the maximum amount of oxygen that can be bound by hemoglobin (Hb) in 100ml of blood when PaO2 is 19.95kPa (150mmHg), PaCO2 is 5.32kPa (40mmHg) and 38°C. The level of CO2max depends on the influence of the quality and quantity of Hb, reflecting the ability of blood to carry oxygen.
The normal blood oxygen capacity is about 8.92mmol/L (20ml%).
1.3 Oxygen content (CO2)
CO2 refers to the actual oxygen-carrying amount of 100ml of blood, including the oxygen physically dissolved in the plasma and the oxygen chemically combined with Hb.
When the PO2 is 13.3kPa (100mmHg), the oxygen in the physical dissolved state in 100ml of plasma is about 0.3ml, and the chemically combined oxygen is about 19ml. Normal arterial oxygen content (CaO2) is about 8.47mmol/L (19.3ml/dl).
Venous blood oxygen content (CvO2) is
5.35-6.24mmol/L (12ml%-14ml/dl). The oxygen content depends on the oxygen
partial pressure and the quality and quantity of Hb.
1.4 oxygen saturation (oxygen saturation, SO2)
SO2 refers to the percentage of Hb bound to oxygen.
SO2 = (oxygen content - physical dissolved oxygen) / oxygen
capacity X 100%
This value is mainly affected by PO2, and the relationship
between the two is the dissociation curve of oxygenated Hb. Normal arterial
oxygen saturation is 93%-98%; venous oxygen saturation is 70%-75%.
1.5 Arterial-venous oxygen difference (A-Vd O2 )
A-Vd O2 is the difference between CaO2 minus CvO2, and the change of the difference mainly reflects the amount of oxygen taken up by the tissue from a unit volume of blood and the ability of the tissue to utilize oxygen.
The oxygen difference between normal arterial blood and mixed venous blood is 2.68-3.57mmol/L (6ml%-8ml%). When the speed of blood flowing through the tissue is significantly slowed down, the oxygen uptake by the tissue from the blood can increase, the oxygen content in the returning venous blood decreases, and the A-Vd O2 increases; on the contrary, the ability of the tissue to utilize oxygen is significantly reduced, and the relationship between Hb and oxygen is reduced.
The oxygen content in the venous blood with
abnormally enhanced affinity increases, and the A-Vd O2 decreases. A decrease
in Hb content can also cause a decrease in A-Vd O2.
1.6 P50
P50 refers to the partial pressure of oxygen when the Hb oxygen saturation is 50% under certain body temperature and blood pH conditions.
P50 represents the affinity of Hb to O2, and the normal value is 3.47-3.6kPa (26-27mmHg).
When the oxygen dissociation curve moves to the right,
the P50 increases, and when the oxygen dissociation curve moves to the left,
the P50 decreases. For example, when the 2,3-DPG concentration in red blood
cells increases by 1mmol/gHb, the P50 will increase by about 0.1kPa.
Type Features
According to the causes of hypoxia and the characteristics of
blood gas changes, simple hypoxia can be divided into four types:
2.1 Hypotonic hypoxia
Hypotonic hypoxia refers to a marked decrease in Pa O2 that results in insufficient oxygen supply to tissues.
When
Pa O2 is lower than 8kPa (60mmHg), it can directly lead to a significant
decrease in CaO2 and SaO2, so hypotonic hypoxia can also be called hypotonic
hypoxemia (hypotonic hypoxemia).
2.1.1 Reasons
Common causes of hypotonic hypoxia are low inspired oxygen
partial pressure, pulmonary dysfunction, and increased venous infusion into
arterial blood.
(1) Low oxygen partial pressure of inhaled gas: hypoxia
caused by inhalation of low oxygen partial pressure gas, also known as
atmospheric hypoxia.
(2) External respiratory dysfunction: caused by pulmonary
ventilation or ventilation dysfunction, called respiratory hypoxia. Common in
various respiratory diseases, respiratory center depression or respiratory
muscle paralysis.
(3) Venous blood shunting into arteries: more common in
congenital heart disease.
2.1.2 Characteristics of blood oxygen changes
i. Due to the low oxygen pressure diffused into the arterial
blood, PaO2 is reduced, and too low PaO2 can directly lead to the reduction of
CaO2 and SaO2;
ii. If Hb has no abnormal changes in quality and quantity,
CO2max is normal;
iii. When PaO2 decreases, 2,3-DPG in red blood cells increases,
so blood SaO2 decreases;
iv. During hypotonic hypoxia, the decrease of PaO2 and blood
SaO2 reduces CaO2;
v. Arterial-venous oxygen difference decreased or changed little. Usually about 5ml of oxygen is used when 100ml of blood flows through the tissue, that is, A-V d O2 is about 2.23mmol/L (5ml/dl).
The driving force of oxygen diffusion from blood to tissue is the oxygen partial pressure difference between the two. When hypotonic hypoxia occurs, PaO2 and CaO2 decrease significantly, which slows down the diffusion rate of oxygen, and the same amount of blood diffuses to the tissue.
The amount of oxygen decreases,
eventually leading to a decrease in A-V d O2 and tissue hypoxia. In the case of
chronic hypoxia, the changes in A-Vd O2 may not be obvious when the tissue's
ability to utilize oxygen is increased to compensate.
2.1.3 Changes in the color of skin and mucous membranes
The average concentration of deoxygenated Hb in normal capillaries is 26 g/L (2.6 g/dl). In hypotonic hypoxia, the concentration of oxygenated Hb in both arterial and venous blood decreases, and the oxygenated Hb in capillaries must decrease, while the concentration of deoxygenated Hb increases.
When the average concentration of deoxygenated Hb in capillaries increases to more than 50g/L (5g/dl) (SaO2 ≤80% ~ 85%), the skin and mucous membranes can appear bluish purple, which is called cyanosis. Cyanosis is prone to occur in chronic hypotonic hypoxia.
Cyanosis is a manifestation of hypoxia, but patients
with hypoxia do not necessarily have cyanosis. For example, blood hypoxia
caused by anemia may not have cyanosis. Similarly, patients with cyanosis can
also have no hypoxia. For example, in patients with polycythemia vera, due to
the abnormal increase in Hb, the deoxygenated Hb content in the capillaries can
easily exceed 50g/L, so it is easy to appear cyanosis without hypoxia symptoms.
2.2 Hemic hypoxia
Hemic hypoxia refers to tissue hypoxia
caused by changes in the amount or quality of Hb, reducing CaO2 or accompanied
by oxygenated Hb-bound oxygen that is not easily released. Hematologic hypoxia
caused by a decrease in the number of Hb, because PaO2 is normal and CaO2 is
decreased, also known as isotonic hypoxemia.
2.2.1 Reasons
(1) Anemia: also known as anemic hypoxia.
(2) Carbon monoxide (CO) poisoning: The combination of Hb and CO can generate carbon oxygen Hb (carboxyhemoglobin, HbCO). Although the binding speed of CO and Hb is only 1/10 of that of O2 and Hb, the dissociation speed of HbCO is only 1/2100 of that of HbO2. Therefore, the affinity of CO and Hb is greater than that of O2 and Hb. 210 times.
When the inhaled gas contains 0.1% CO, 50% of Hb in the blood can be converted to HbCO, so that a large amount of Hb loses its oxygen-carrying function.
CO can also inhibit glycolysis in red blood cells, reducing the production of 2,3-DPG, The oxygen dissociation curve shifts to the left, and it is difficult for HbO2 to release the bound oxygen; the O2 bound in HbCO is also difficult to release.
Severe tissue hypoxia is caused by the loss of HbCO to carry O2 and hinder the dissociation of O2. There is about 0.4% HbCO in normal human blood. When the air contains 0.5% CO, the HbCO in the blood can reach as high as 70% in only 20 to 30 minutes.
Organs
with high metabolism, high oxygen demand, and fewer vascular anastomotic
branches are more vulnerable to CO poisoning.
(3) Methemoglobinemia: When nitrite, perchlorate, sulfonamide, etc. are poisoned, a large amount (20% to 50%) of Hb in the blood can be converted into methemoglobin (methemoglobin, HbFe 3+ OH).
The formation of high iron Hb is due to the oxidation of divalent iron in Hb to trivalent iron under the action of oxidants, so it is also called denatured Hb or hydroxylated Hb.
The Fe3+ in the high iron Hb loses its ability to carry oxygen due to its strong bond with the hydroxyl group. In addition, when part of the Fe2+ in the Hb molecule is oxidized to Fe3+, the Fe2+ on the remaining pyrrole ring has a higher affinity for O2, and the oxygen dissociation curve shifts to the left, and the high iron Hb is not easy to tolerate. The bound oxygen is released, aggravating tissue hypoxia.
Patients may experience symptoms such as headache, weakness, coma, dyspnea, and tachycardia due to hypoxia.
It is common
clinically to eat a lot of freshly pickled pickles or spoiled vegetables.
Because they contain a lot of nitrate, nitrate is reduced to nitrite by
gastrointestinal bacteria and absorbed through the intestinal mucosa, causing
high iron Hb hyperemia. The patient's skin and mucous membranes (such as the
lips) appear bluish-gray, also known as enterogenous cyanosis.
In physiological state, there is also a small amount of high iron Hb in the blood, but it can be reduced to Fe2+ by reducing agents such as NADH, vitamin C, reduced glutathione, etc. in the body, so that the content of high iron Hb in normal blood is limited to the total amount of Hb. 1% to 2%.
MetHbemia can also be seen in a type of HbM hereditary MetHbemia. This disease
is due to a 58 group → tyrosine mutation, where tyrosine occupies the ligand
position of the Fe atom of heme, making it a stable high iron state, and the
patient has symptoms of cyanosis and secondary polycythemia.
(4) Abnormal increase in the affinity of Hb and oxygen: seen in the input of a large amount of stock blood or alkaline liquid, and also in some hemoglobinopathies. The low content of 2,3-DPG in the red blood cells of the bank blood shifts the oxyhemoglobin dissociation curve to the left.
The
mutation of the gene, when the 92nd arginine of the a chain is replaced by
leucine, the affinity of Hb to O2 is several times higher than normal. times.
2.2.2 Characteristics of blood oxygen changes
When anemia causes hypoxia, due to the normal external
respiratory function, PaO2 and SaO2 are normal, but due to the decrease in the
number or nature of Hb, the decrease in oxygen capacity leads to a decrease in
CaO2.
In CO poisoning, the changes in blood oxygen are basically
the same as those in anemia. However, CO2max can be normal when detected in
vitro, because the blood sample is fully equilibrated with oxygen in vitro. At
this time, O2 has completely competed to replace CO in HbCO to form oxygenated
Hb, so blood CO2max can be normal.
When blood is hypoxia, when blood flows through capillaries,
due to insufficient total amount of HbO2 in blood and rapid decrease of PO2,
the diffusion power and speed of oxygen are also rapidly reduced, so A-V d O2
is lower than normal.
Hematological hypoxia caused by the increased affinity of Hb
and O2 is special, and its PaO2 is normal; CaO2 and SaO2 are normal. Because Hb
has a high affinity with O2, the bound oxygen is not easy to release and leads
to tissue hypoxia, so PvO2 increases; CvO2 and Sv O2 is elevated, A-V d O2 is
less than normal.
2.2.3 Color changes of skin and mucous membranes
When pure Hb is reduced, the oxyhemoglobin is reduced, and the reduced Hb in the capillaries of the patient does not reach the threshold of cyanosis, so the color of the skin and mucous membranes is relatively pale.
HbCO itself has a particularly bright red color. In severe hypoxia, the skin and mucous membranes are pale due to vasoconstriction of the skin.
In the case of high iron Hb hyperemia, due to the increase in the content of high iron Hb in the blood, the skin and mucous membranes of the patient appear dark brown or blue. Purple.
When the affinity between Hb and O2 is simply increased, the
amount of deoxygenated Hb in the capillaries is less than normal, so the
patient's skin and mucous membranes have no cyanosis.
2.3 Circulatory hypoxia
Circulatory hypoxia (circulatory
hypoxia) refers to hypoxia caused by decreased tissue blood flow and reduced
tissue oxygen supply, also known as hypokinetic hypoxia. Circulating hypoxia
can also be divided into ischemic hypoxia and congestive hypoxia. Ischemic
hypoxia is due to insufficient arterial blood supply; congestive hypoxia is due
to obstruction of venous return.
2.3.1 Reasons
The cause of circulatory hypoxia is reduced blood flow, which
can be divided into two types i.e. systemic and local.
(1) Systemic decrease in blood flow
(2) Local blood flow decrease
2.3.2 Characteristics of blood oxygen changes
In simple circulatory disorders, the blood oxygen volume is normal; PaO2 is normal, CaO2 is normal, and SaO2 is normal.
Due to the slow blood flow, the time for blood to flow through the capillaries is prolonged, so that the amount of oxygen per unit volume of blood diffused to the tissue increases, and the C v O2 decreases, so the A-Vd O2 blood oxygen difference also increases.
Decreased oxygen supply to cells can also cause tissue hypoxia.
In localized circulatory hypoxia, blood oxygen changes can be basically normal.
2.3.3 Color changes of skin and mucous membranes
Due to the low CvO2 and PvO2 of venous blood, deoxygenated Hb
in capillaries can exceed 50g/L, which can cause skin and mucous membrane
cyanosis.
2.4 Tissue hypoxia Tissue hypoxia (histogenous hypoxia)
refers to the hypoxia caused by the disturbance of oxygen utilization by
tissues and cells.
2.4.1 Reasons
(1) Inhibit cellular oxidative phosphorylation
Iron in cytochrome molecules conducts electron transfer via a reversible redox reaction, a key step in cellular oxidative phosphorylation.
Taking cyanide as an example, when various inorganic or organic cyanides such as: HCN, KCN, NaCN, NH4CN and organic derivatives of hydrocyanic acid (mostly present in the kernels of apricot, peach and plum) are digested after entering the body through the tract, respiratory tract, and skin. CN- can quickly combine with intracellular oxidative cytochrome oxidase ferric iron to form ferric cyanide cytochrome oxidase (CNˉ+ Cytaa 3Fe3+ → Cyt aa 3Fe3+ -CNˉ ), which loses the electron accepting effect. The ability to interrupt the respiratory chain, causing tissue cells to use oxygen barriers. 0.06gHCN can cause death in humans.
High concentrations of CO can also combine with Fe2+ of oxidative cytochrome oxidase aa to block the respiratory chain. Poisoning such as hydrogen sulfide, arsenic, and methanol prevents cellular oxidative processes by inhibiting the activity of cytochrome oxidase.
Antimycotin A and
phenformin can inhibit the transfer of electrons from cytochrome b to
cytochrome c, block the respiratory chain and lead to tissue toxicity and
hypoxia.
(2) Mitochondrial Damage
Causes of mitochondrial damage include: strong radiation,
bacterial toxins, heat stroke, uremia, etc. Mitochondrial damage can lead to
impaired oxygen utilization and reduced ATP production in tissue cells.
(3) Respiratory Enzyme Synthesis Disorder
Vitamin B1, B2, nicotinamide, etc. are cofactors of coenzymes
in the body's energy metabolism, and the lack of these vitamins leads to
obstacles to tissue cells' oxygen utilization and ATP generation.
2.4.2 Characteristics of blood oxygen changes
During tissue hypoxia, the blood oxygen volume is normal, and PaO2, CaO2, and SaO2 are generally normal. Due to the obstacle of oxygen utilization by tissue cells (internal respiratory disorder), Pv O2 , C v O2 , S v O2 are increased, and (A-V) d O2 is less than normal.
The color of the
patient's skin and mucous membranes is often bright red or rose red because the
amount of oxygenated Hb in the capillaries is higher than normal.
The most common clinical hypoxia is mixed hypoxia. For
example, pulmonary heart disease can cause respiratory hypoxia due to pulmonary
dysfunction, and circulatory hypoxia can occur in cardiac insufficiency.
|
The
characteristics of blood oxygen changes in various types of Hypoxia |
||||
|
Type of hypoxia |
Arterial partial pressure
of oxygen |
Blood oxygen capacity |
Arterial hemoglobin oxygen
saturation |
Poor arterial and venous
oxygen content |
|
Hypotonic hypoxia |
Decline |
Constant |
Decline |
Decrease or unchanged |
|
Blood hypoxia |
Constant |
Decrease or unchanged |
Constant |
Decline |
|
Cyclic hypoxia |
Constant |
Constant |
Constant |
Rise |
|
Tissue hypoxia |
Constant |
Constant |
Constant |
Decline |
Function Change
Oxygen is taken in by the body and transported through the blood to tissues, where it is finally sensed and utilized by cells. Therefore, the essence of hypoxia is a response and adaptive change of cells to the hypoxic state.
When acute severe hypoxia occurs, the cellular changes are dominated by mitochondrial energy metabolism disorders (including tissue toxic hypoxia).
In chronic mild hypoxia cells, the compensatory regulation of oxygen
receptors is the main factor.
3.1 Compensatory changes
3.1.1 Changes in cellular energy metabolism during hypoxia
(1) Enhanced anaerobic glycolysis: When Pa O2 decreases, and P O2 around mitochondria is lower than 0.04-0.07kPa, oxygen as the final electron acceptor in the aerobic oxidation process is deficient, and mitochondrial aerobic metabolism is hindered, ATP production decreased, cytoplasmic ADP increased.
The increase of ADP in the cytoplasm can enhance the process of
phosphofructokinase and glycolysis, and to a certain extent, it can compensate
for the lack of energy in cells, but the acidic products increase.
(2) Enhanced ability to utilize oxygen: In chronic chronic
and mild hypoxia, the number of mitochondria in cells increases, and the
activity and content of biological oxidoreductases (such as succinate dehydrogenase,
cytochrome oxidase) increase, which enables cells to utilize oxygen. Oxygen
capacity is enhanced.
3.1.2 Oxygen sensitivity regulation and adaptive changes of cells
(1) Excitation of chemoreceptors
(2) Hemeprotein (hemeprotein) sensing regulation: hemoglobin refers to a class of proteins containing porphyrin ring ligands, such as hemoglobin, cytochrome aa3, P450, coenzyme II (NADPH) oxidase containing cytochrome b 558, etc.
There are two types of sensory adjustment:
i. Conformational change When O2 binds to Fe2+ in the center of
the heme molecule, it causes Fe2+ to translocate to the plane of the porphyrin
ring, and vice versa. This conformational change may affect the function of
heme proteins. For example: CO combines with Fe2+ of oxidative cytochrome
oxidase aa, so that oxidative cytochrome oxidase loses the role of electron
transfer.
ii. The messenger molecule NADPH oxidase can combine with O2 in the surrounding environment of cells, and convert O2 into O2-, and then generate H2O2.
H2O2 is converted into hydroxyl radical (OH-) through Feton reaction for oxygen signal transduction.
Under normal conditions, the intracellular H2O2 concentration is relatively high, which inhibits the expression of hypoxia-sensitive genes. In hypoxia, the generation of H2O2 and OH- in cells decreases, and the oxidative conversion of reduced glutathione (GSH) into oxidized glutathione (GSSG) is inhibited, resulting in an increase in the reduced thiol group of some proteins, thereby making some transcription factors.
The conformation is altered to promote the transcriptional expression
of hypoxia-sensitive genes.
3.1.3 HIF-1 sensory regulation
Studies have shown that HIF-1 (hypoxia induced factor-1) is a crucial transcription factor controlled by changes in oxygen concentration.
In
the nucleus, HIF-1 acts as the promoter of hypoxia-sensitive genes and binds to
the hypoxia response element (HRE, 5-RCGTG-3) of target genes to initiate gene
transcription and protein translation.
3.1.4 Increased red blood cell adaptation
People living in high altitudes and people with chronic hypoxia for a long time can increase red blood cells to 6×106/㎜ 3 and Hb to 21g/dl.
The mechanism of its increase is that when hypoxia is present, hypoxic blood can stimulate the juxtaspheric cells to increase the production of erythropoietin (erythropoiesis-stimulating factor, EPO).
EPO can stimulate the differentiation, proliferation and maturation of RBC unidirectional stem cells into original RBC. in addition. EPO can promote the synthesis of Hb and the entry of reticulocytes into the blood, increase the red blood cells and Hb in the blood, and improve the blood oxygen capacity in the blood.
Ultimately, it
improves the blood's ability to carry oxygen and increases the oxygen content,
thereby enhancing the O2 supply to tissues and organs.
3.1.5 Myoglobin (Mb) increase
Since the affinity of Mb with oxygen is greater than that of Hb, for example, when the oxygen partial pressure drop is 10mmHg, the oxygen saturation of Hb is about 10%, while the oxygen saturation of Mb can reach 70%.
When the tissue oxygen partial pressure is further reduced, Mb can release a
large amount of oxygen for tissue and cell utilization. Increased Mb may have a
role in storing oxygen.
3.2 Cell damage
Hypoxic cell damage (hypoxic cell damage) is
often a decompensatory change that occurs during severe hypoxia. It is mainly
manifested as damage to the cell membrane, mitochondria and lysosomes.
3.2.1 Cell membrane changes
The decrease in cell membrane potential often precedes the
decrease in intracellular ATP content. The reason for the decrease in membrane
potential is that the permeability of the cell membrane to ions increases,
resulting in ion concentration difference passing through the cell membrane,
followed by sodium influx, potassium outflow, calcium influx and cellular A
series of changes such as edema.
(1) Na+ influx: increase the intracellular Na+ concentration and activate the Na+-K+ pump, pump out intracellular Na+ and consume too much ATP, which in turn will promote mitochondrial oxidative phosphorylation and aggravate cellular hypoxia.
Too high intracellular Na+ concentration will
inevitably lead to cell edema accompanied by the increase of water entering
into the cell. Cell edema is the basis of mitochondrial and lysosome swelling.
(2) K+ efflux: Due to the dysfunction of the Na+-K+ pump,
extracellular K+ cannot be pumped into the cytoplasm, the lack of K+ in the
cell leads to anabolic disorders, the production of various enzymes is reduced
and further affects the production of ATP and the ion pump. Function.
(3) Ca2+ influx: The difference between intracellular and extracellular Ca2+ concentration is about 1000 times, and the maintenance of low intracellular Ca2+ concentration depends on the function of Ca2+ pump on the membrane.
In severe hypoxia, due to the reduction of ATP generation, the function of Ca2+ pump on the membrane is reduced, the efflux of Ca2+ in the cytoplasm and the uptake of Ca2+ in the sarcoplasmic reticulum are hindered, so that the concentration of Ca2+ in the cytoplasm increases.
The increase of intracellular Ca2+ and its entry into the mitochondria inhibits the function of the respiratory chain. Ca2+ and calmodulin activate phospholipase, decompose membrane phospholipids, cause lysosomal damage and the release of hydrolase, and cell autolysis. intracytoplasmic
Excessive Ca2+ concentration can convert xanthine dehydrogenase into xanthine oxidase, increase the formation of free radicals, and aggravate cell damage.
3.2.2 Changes in mitochondria
Hypoxia can damage mitochondria, and mitochondrial damage can lead to hypoxia, both of which are mutually causal.
The reason for mitochondrial damage caused by hypoxia is that severe hypoxia can significantly inhibit mitochondrial respiration and oxidative phosphorylation process, and reduce ATP generation.
Severe hypoxia for a long time can reduce or disappear
mitochondrial matrix particles, matrix electrons Increased density, dilation of
the ridge cavity, swelling, disintegration of the ridge, rupture of the
adventitia, etc.
3.2.3 Changes in lysosomes
In hypoxia, the increased production of lactic acid due to enhanced glycolysis and insufficient fat oxidation increase ketone bodies, resulting in acidosis.
The decrease of pH and the increase of intracytoplasmic calcium increase the activity of phospholipase, the phospholipids of the lysosomal membrane are decomposed, the permeability of the membrane is increased, the lysosome swells, ruptures and releases a large amount of lysosomal enzymes, which in turn leads to the formation of cell and dissolution and necrosis of surrounding tissue.
Intracellular edema and the effects of free
radicals also participate in the mechanism of lysosomal damage.
Metabolic Changes
The effect of hypoxia on organs depends on the degree, speed and duration of hypoxia and the functional metabolic state of the body.
Chronic
mild hypoxia mainly causes compensatory response of organs; acute severe
hypoxia often leads to decompensation and dysfunction of organs, and even
causes irreversible damage to vital organs, leading to the death of the body.
4.1 Changes in the respiratory system
4.1.1 Compensatory
responses
4.1.1.1 Deeper and faster breathing
4.1.1.2 Increased thoracic respiratory movement
The increase in respiratory movement mainly caused by
hypoxemia increases the negative intrathoracic pressure, promotes the increase
of venous return, increases cardiac output and pulmonary blood flow, and is
conducive to oxygen uptake and transport.
Changes in pulmonary ventilation caused by hypotonic hypoxia
are related to the duration of hypoxia.
The air PO2 of the 4000m plateau is 100mmHg, and the alveolar air PO2 is about 55mmHg. Therefore, under the hypoxic perception of chemoreceptors, pulmonary ventilation increases immediately, and due to thin air, PCO2 is also low, CO2 exhalation increases (respiratory alkalosis and hypoxemia occur), PaCO2 decreases, and the center of the medulla oblongata is reduced.
The stimulation of chemoreceptors limits the increase of lung ventilation, so the early lung ventilation is only 65% higher than sea level; after a few days, HCO3- is compensatively eliminated by the kidneys, and HCO3- in the cerebrospinal fluid also enters the blood through the blood-brain barrier.
The pH in the brain tissue is gradually returned to normal, the stimulation inhibition of the central chemoreceptors in the medulla oblongata is gradually relieved, and the ventilation volume of the lung can be increased to 5-7 times of sea level.
The lung ventilation volume of long-term residents
gradually drops to only higher than the sea level. 15% higher, which may be
related to the decreased sensitivity of peripheral chemoreceptors to hypoxia.
This is also a chronic adaptation process, because for every 1L increase in
lung ventilation, the oxygen consumption of the respiratory muscles increases
by 0.5ml, so the increase in long-term breathing exercise is obviously
unfavorable to the body.
4.1.2 Respiratory dysfunction
High altitude pulmonary edema (HAPE), manifested as dyspnea,
cough, bloody foamy sputum, moist rales in the lungs, and cyanosis of the skin
and mucous membranes. Its pathogenesis is related to the following factors:
i. Hypoxia causes peripheral vasoconstriction, increased blood
return to the heart and increased pulmonary blood volume, coupled with hypoxic
pulmonary vasoconstriction response to increase pulmonary blood flow
resistance, resulting in pulmonary hypertension.
ii. Intensity of pulmonary vasoconstriction causes uneven
distribution of pulmonary blood flow. In the parts where pulmonary
vasoconstriction is mild or does not occur, alveolar capillary blood flow
increases and hydrostatic pressure increases, causing pressure pulmonary edema.
iii. High intrapulmonary blood pressure and high flow velocity
increase the shear stress on microvessels (the component force of the force
acting on the blood vessels in the direction parallel to the vessel wall) by
the flowing blood.
iv. The permeability of the microvascular wall of the lung is
increased. For example, vasoactive substances such as complement C3a, LTB4 and
TXB2 may lead to the damage and permeability of microvascular endothelial
cells.
Pulmonary edema affects the ventilation function of the lung,
which can further decrease PaO2 and aggravate hypoxia. Too low PaO2 can
directly inhibit the respiratory center, resulting in respiratory depression
and reduced pulmonary ventilation, leading to respiratory failure.
4.2 Changes in the circulatory system 4.2.1 Increased cardiac output
The main mechanisms leading to increased cardiac output are:
i. Rapid heart rate: When inhaling air containing 8% O2, the
heart rate can be doubled. It is believed that the increased heart rate is
likely to be the stimulation of lung stretch receptors by lung expansion caused
by increased ventilation, which reflexively inhibits the effect of the vagus
nerve on the heart.
ii. Enhanced myocardial contractility: As a stressor, hypoxia
can increase sympathetic nerve excitation and catecholamine release, and act on
cardiac β-adrenergic receptors to increase heart rate and enhance myocardial
contractility.
iii. Increased venous return: thoracic movement and cardiac
activity are enhanced during hypoxia, the negative pressure in the thoracic
cavity increases, venous return increases, and cardiac output increases.
4.2.2 Blood redistribution
During acute hypoxia, the skin and abdominal viscera are excited by sympathetic nerves, and the vasoconstrictor effect is dominant, which makes vasoconstriction; while cerebral vasoconstriction is not obvious.,
Blood flow increases under the vasodilator effect of glycosides and PGI 2 ,
etc. This change in systemic blood flow distribution is obviously beneficial to
ensure the supply of oxygen to vital organs of life.
4.2.3 Pulmonary vasoconstriction (pulmonary blood vessels respond to hypoxia in the opposite way to body blood vessels)
i. Sympathetic nerve excitation causes pulmonary vasoconstriction. Sympathetic nerve excitability caused by acute hypoxia can act on α1 receptors in pulmonary blood vessels to cause vasoconstriction.
Changes in the distribution of receptors in pulmonary vascular smooth muscle
during chronic hypoxia: α1 receptors increase and β receptor density decreases,
resulting in enhanced pulmonary vasoconstriction.
ii. the role of humoral factors to pulmonary vasoconstriction. Mast cells, alveolar macrophages, vascular endothelial cells and vascular smooth muscle cells in the lung tissue can release various vasoactive substances, such as: mast cells degranulate to release histamine, VEC to release PGI 2, and ET increases to cause pulmonary vasoconstriction.
In the
process of vasoconstriction, the increase of vasoconstrictor substances plays a
leading role, and the increase of vasodilator substances plays a feedback
regulation role.
iii. The direct feeling of vascular smooth muscle to hypoxia. As described in the section on cellular metabolism and functional changes during hypoxia, hypoxia can directly close the oxygen-sensitive potassium channels on the membrane of pulmonary vascular smooth muscle cells, resulting in decreased intracellular K+ efflux, decreased membrane potential, increased cell excitability, and increased cell excitability.
Accelerated metabolism and
enhanced influx of extracellular Ca2+ eventually led to pulmonary
vasoconstriction.
In addition to pulmonary vasoconstriction leading to
pulmonary hypertension, chronic hypoxia also has hypertrophy and thickening of
smooth muscle in the middle of the pulmonary vascular wall, as well as
hyperplasia of elastic fibers and collagen fibers, which reduces the diameter
of blood vessels and increases blood flow resistance.
4.2.4 Capillary hyperplasia
Long-term mild hypoxia of tissue cells can increase the
synthesis and release of VGEF by the cells through the hypoxia sensing of
HIF-1a, and the capillaries proliferate in hypoxic tissues (see changes in cell
metabolism and function during hypoxia). This phenomenon is more pronounced in
the brain, hypertrophic myocardium, solid tumors, and skeletal muscle, where
capillary hyperplasia is present.
4.3 Changes in the blood system Hypoxia can enhance bone marrow
hematopoiesis and shift the oxyhemoglobin dissociation curve to the right.
4.3.1 Polycythemia
4.3.2 Right shift of oxyhemoglobin dissociation curve
In hypoxia, 2,3-DPG in red blood cells increases, resulting
in a rightward shift of the oxygenated Hb dissociation curve, and Hb easily
releases the bound oxygen for tissue utilization.
(1) There are two reasons for the increased production of
2,3-DPG in erythrocytes:
i. In hypotonic hypoxia, oxygenated Hb decreases and deoxygenated Hb increases. The former has a small central hole and cannot bind 2,3-DPG; the latter has a larger central hole and can bind 2,3-DPG.
When
deoxy-Hb increases, the free 2,3-DPG in erythrocytes decreases, and the
inhibitory effect of 2,3-DPG on phosphofructokinase and diphosphoglycerate
mutase (DPGM) weakens, thereby enhancing glycolysis , 2,3-DPG generation
increased.
ii. Hypotonic hypoxia is caused by compensatory pulmonary hyperventilation causing respiratory alkalosis, and a large amount of deoxygenated Hb existing in erythrocytes during hypoxia is slightly alkaline, which increases the pH in erythrocytes, thereby activating phosphofructokinase and inhibiting 2, 3-DPG phosphatase (2,3-DPG phosphatase, 2,3-DPGP) activity. The former enhances glycolysis and increases the synthesis of 2,3-DPG; the latter reduces the decomposition of 2,3-DPG.
(2) The mechanism by which the increase of 2,3-DPG shifts the
dissociation curve of oxygenated Hb to the right is:
i. The steric configuration of deoxygenated Hb combined with
2,3-DPG is relatively stable, and it is not easy to combine with oxygen;
ii. 2,3-DPG is an organic acid that cannot penetrate
erythrocytes, and its increase can reduce the pH in erythrocytes. The decrease
in pH shifts the oxygenated Hb dissociation curve to the right through the Bohr
effect. However, when Pa O2 is lower than 8kPa, the rightward shift of the
oxygen dissociation curve can significantly affect the uptake of oxygen by the
pulmonary blood.
4.3.3 Hemoglobin phenotype reconstruction
4.4 Changes in the central nervous system The central nervous system is the organ most sensitive to hypoxia because the brain has a very high demand for oxygen. Brain weight is only 2% of body weight, cerebral blood flow accounts for 15% of cardiac output, and cerebral oxygen consumption accounts for 23% of total oxygen consumption. Therefore, the brain is very sensitive to hypoxia.
Clinically, the brain is completely hypoxic for 5-8 minutes.
Irreversible damage can occur later.
Acute hypoxia can cause headache, emotional agitation, decreased or lost thinking, memory, judgment, and motor incoordination. Severe hypoxia can cause morphological changes such as cell swelling, degeneration, necrosis and cerebral interstitial edema in the brain tissue, which is related to the increase of cerebral microvascular permeability caused by hypoxia and acidosis. These injuries often occur within minutes of hypoxia, and irreversible.
Cerebral vasodilation, brain cells and interstitial edema can
increase intracranial pressure, resulting in headache, vomiting, restlessness,
convulsions, coma, and even death. Chronic hypoxia is prone to fatigue,
lethargy, inability to concentrate and other symptoms.
The mechanism of extremely severe hypoxia that can lead to
coma and death is due to the decrease of nerve cell membrane potential and the
decrease of neurotransmitter synthesis; the energy metabolism disorder of brain
cells, the decrease of ATP, and the increase of cell membrane permeability;
acidosis, the increase of intracellular free Ca2+, the increase of soluble Ca
Factors such as the release of enzymatic enzymes and cell edema lead to
dysfunction of the central nervous system.
The pathogenesis of the so-called high altitude cerebral edema (HACE) may be related to the following factors in addition to cerebral vasodilation and increased cerebral blood flow caused by hypoxia.
1) Cerebral cell edema
2) Impaired blood-brain barrier function
3) Thrombosis in cerebral veins, further aggravating the formation of cerebral edema
Therapeutic Methods
1. Physical oxygen production
Through the pressure swing adsorption of molecular sieves,
the oxygen and nitrogen in the air are directly separated at room temperature
to extract high-purity medical oxygen, which can achieve continuous and
uninterrupted oxygen supply.
This method is suitable for middle-aged and elderly people,
people with poor physical fitness, pregnant women, college entrance examination
students, obese people and other people with different degrees of physiological
hypoxia. It can also eliminate fatigue and restore physical function after heavy
physical or mental exhaustion. to a certain effect.
2. Chemical oxygen production
Using the principle of chemical reaction of chemical
substances to generate oxygen, manganese dioxide and potassium chlorate are
mostly used.
The oxygen supply method of the oxygen generator is mostly
used for the symptoms of environmental hypoxia to improve the impact of the
hypoxic environment of the external environment on the body.
You can choose different methods to improve your hypoxia according
to your own situation.
What is Liquid oxygen therapy?
Use biologically available oxygen solution, bypass the respiratory tract, and directly absorb oxygen through mucous membranes, digestive tract, skin, etc.
This method is especially suitable for patients with respiratory diseases, who need oxygen supplementation due to hypoxia, but are inconvenient for oxygen supplementation, such as high altitude tourists.
This is a milestone in oxygen supplementation technology, breaking through the bottleneck of life that oxygen supplementation can only be breathed through the lungs.
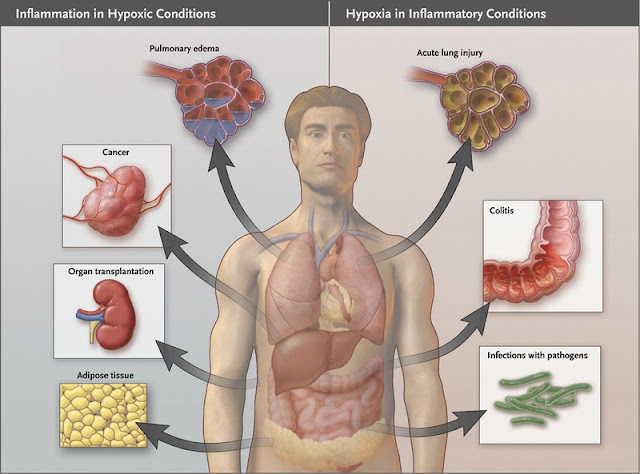
Can hypoxia induce infalmmation?
Hypoxia can induce inflammation and inflamed lesions often become severely hypoxic.
As a result of the steep oxygen gradient between the anaerobic intestinal lumen and the metabolically active lamina propria mucosae, intestinal epithelial cells become normally hypoxic.
Do mountain climbers suffer from Hypoxia?
Of course mountaineers can suffer from hypoxia as it is concerned to breathing, and breathing is difficult while climbing and also in high altitudes.
Author's Bio
Education: MBBS, MD
Occupation: Medical Doctor
Specialization: Community Medicine, General Surgery, Natural Treatment
Experience: 18 Years as a Medical Practitioner
[[SHARE in Public Health Interest>>





Comments
Post a Comment